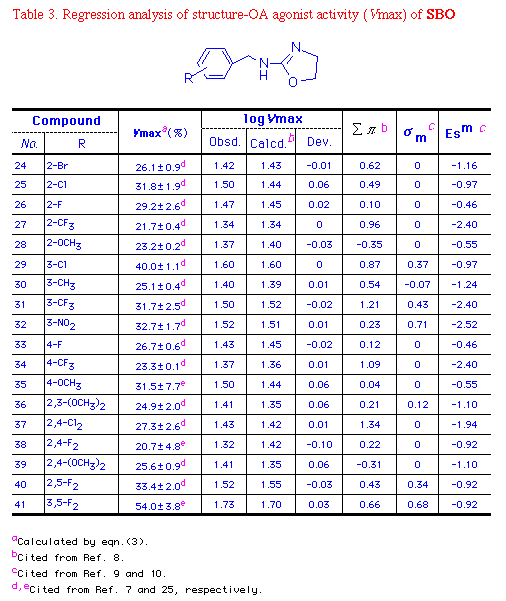
3.2. QSAR of SBO
logVmax = 1.485(±0.022) + 0.096(±0.030) 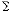 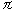 + 0.353(±0.046) + 0.353(±0.046)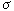 m + 0.098(±0.021)Es ............ (3) m + 0.098(±0.021)Es ............ (3)
where n=18, r=0.9134, s=0.0453, and F=23.489.
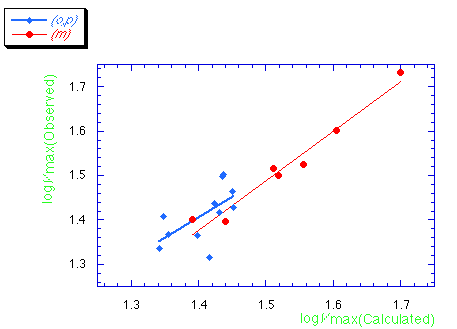
Chart 2. Observed logVmax vs Calculated value from eqn.
(3) for QSAR of SBOs
The correlation of calculated logVmax from eqn. (3) and
observed value from bioassay was examined and shown in Chart 2.
According to eqn. (3) , the electronic nature of a m-substituent
was the most important followed by the steric and hydrophobic
effects for SBO. The positive 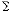 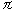 and
and 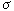 m terms mean that
the more hydrophobic and the more electron-withdrawing the
substituent, the greater the activity. 3,5-F2
derivative 41 had the highest Vmax, followed by
3-Cl (29) and 2,5-F2 (40) derivatives
for stimulating adenylate cyclase prepared from thoracic nerve
cords of P. americana (Table 3). m terms mean that
the more hydrophobic and the more electron-withdrawing the
substituent, the greater the activity. 3,5-F2
derivative 41 had the highest Vmax, followed by
3-Cl (29) and 2,5-F2 (40) derivatives
for stimulating adenylate cyclase prepared from thoracic nerve
cords of P. americana (Table 3).
pKa = 2.528(±0.675) + 1.962(±0.446)logVmax
......... (4)
where n=8, r=0.8739, s=0.1613, and F=19.386.
According to eqn. (4), the positive logVmax term means
that the larger Vmax value, the higher pKa value.
The observation that relationship between Vmax and pKa
for SBOs is apparently not matched with that for SBTs
implies there may be remarkable variations in the
agonist-receptor interaction mechanism for these types of
compounds. In addition, the presence of alkyl groups at the p-position
or of an alkyl group at m-position of SBO seemed
particularly important (Table 4).
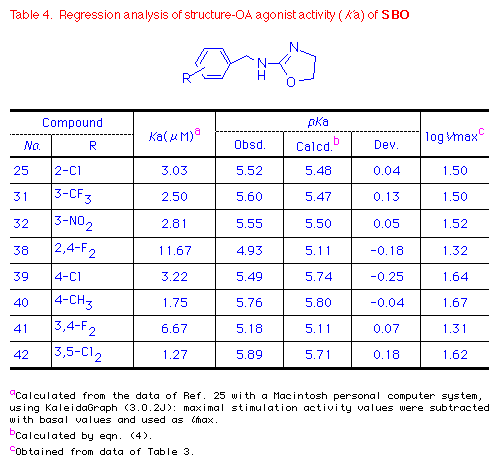
Oxazoline 3-Cl-SBO (29) was more potent in terms of
both Ka (0.52  M)
and Vmax (40% relative to OA) than its thiazoline
derivative (Ka=1.61 M)
and Vmax (40% relative to OA) than its thiazoline
derivative (Ka=1.61  M, Vmax=23% relative to OA). Similarly, 2,5-F2-SB0
(40) (Ka=0.55 M, Vmax=23% relative to OA). Similarly, 2,5-F2-SB0
(40) (Ka=0.55  M, Vmax=33%) were more potent than thiazoline 16
(Ka=3.5 M, Vmax=33%) were more potent than thiazoline 16
(Ka=3.5 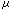 M, Vmax=27%).
2-Cl- (25), 3-CF3- (31), 4-Cl-, 4-F-(33),
4-CH3-, 4-OCH3- (36), 2,3-(OCH3)2-,
3-Cl,4-F-, 3,4-F2-, and 3,5-F2-SBO (41)
had higher Vmax values (32, 32, 44, 27, 47, 32, 25, 16,
21, and 54%, relative to OA) than thiazolines [24, 26 (3-CF3)
(8), 22 (4-Cl) (9), 20, 26, 29 (4-OCH3)
(11), 8, 12 (3-Cl,4-F) (21), 19 (3,4-F2)
(22), and 33% (3,5-F2) (35) relative to
OA] suggesting that SBOs are more potent than their
thiazoline derivatives (SBTs). M, Vmax=27%).
2-Cl- (25), 3-CF3- (31), 4-Cl-, 4-F-(33),
4-CH3-, 4-OCH3- (36), 2,3-(OCH3)2-,
3-Cl,4-F-, 3,4-F2-, and 3,5-F2-SBO (41)
had higher Vmax values (32, 32, 44, 27, 47, 32, 25, 16,
21, and 54%, relative to OA) than thiazolines [24, 26 (3-CF3)
(8), 22 (4-Cl) (9), 20, 26, 29 (4-OCH3)
(11), 8, 12 (3-Cl,4-F) (21), 19 (3,4-F2)
(22), and 33% (3,5-F2) (35) relative to
OA] suggesting that SBOs are more potent than their
thiazoline derivatives (SBTs).
Next
page /Previous page /Table of Contents
|
